Like every dog owner, you want to avoid fleas on your dog and in your home. And you don’t want your dog picking up ticks and risking tick-borne illnesses like lyme disease, skin irritation, or rocky mountain spotted fever.
Wouldn’t it be convenient to just give your dog a tasty chew every month or 3 months to protect her from both fleas and ticks? Well, in the last few years, some new preventives for dogs let you do just that.
Great! Problem solved… or is it?
No, it’s not! If you got excited about the idea of a longer term chewable flea and tick medicine … stop right there!
These tick preventatives have a dark side and higher risk than you might expect. Keep reading to learn about their sometimes deadly side effects and active ingredients.
What Are Chewable Flea And Tick Preventives?
There are three drugs in the category of chewable flea and tick preventives: Nexgard, Bravecto and Simparica.
Nexgard (active ingredient afoxolaner) and Bravecto (fluralaner) were approved in the US in late 2013 and early 2014. Simparica (sarolaner) came out in March 2016.
The dosing schedules are once a month for Nexgard and Simparica and once every three months for Bravecto.
How Oral Flea And Tick Preventives Work
These flea treatments are oral medications. The drugs come in a soft chew that your dog can eat like a treat. After your dog takes the chew, the drugs circulate in the blood, and when a flea or tick bites your dog, it’s exposed to the chemical. This will kill fleas and ticks.
All three drugs are pesticides that work by attacking the nervous system of the fleas and ticks, causing death. The excerpts below from the manufacturers’ Prescribing Information explain the mode of action of these drugs (we added some bold to highlight the important parts) …
Nexgard (afoxolaner)
“Afoxolaner is a member of the isoxazoline family, shown to bind at a binding site to inhibit insect and acarine ligand-gated chloride channels, in particular those gated by the neurotransmitter gamma-aminobutyric acid (GABA), thereby blocking pre- and post-synaptic transfer of chloride ions across cell membranes. Prolonged afoxolaner-induced hyperexcitation results in uncontrolled activity of the central nervous system and death of insects and acarines.“
Bravecto (fluralaner)
“Fluralaner is for systemic use and belongs to the class of isoxazoline-substituted benzamide derivatives. Fluralaner is an inhibitor of the arthropod nervous system. The mode of action of fluralaner is the antagonism of the ligand-gated chloride channels (gamma-aminobutyric acid (GABA)-receptor and glutamate-receptor).”
Simparica (sarolaner)
“The active substance of SIMPARICA, sarolaner, is an acaricide and insecticide belonging to the isoxazoline group. Sarolaner inhibits the function of the neurotransmitter gamma aminobutyric acid (GABA) receptor and glutamate receptor, and works at the neuromuscular junction in insects. This results in uncontrolled neuromuscular activity leading to death in insects or acarines.“
So, these chewable drugs work by destroying the insects’ nervous systems. If it’s deadly for fleas and ticks, how might it affect a dog? Remember … these drugs stay in your dog’s bloodstream for extended periods of time.
That means … once your dog takes one of these drugs, if she has any side effects, you can’t remove the drug from her body.
What Adverse Drug Event Reports Say
Dog owners often ask, “What is the safest flea and tick prevention for dogs?” Well, it’s not these chewable drugs.
Every drug has side effects and so we wanted to review the Adverse Drug Event Reports submitted to the FDA. But the FDA has made it near-impossible to find these ADE reports on their database. Luckily, thanks to veterinarian Dr Elizabeth Carney, we were able to find some information.
Back in November 2014, when Nexgard and Bravecto were first introduced, Dr Carney wrote about the new chewable drugs on her blog. And then she started receiving comments from people whose dogs were already experiencing side effects after taking these preventives. She wanted to find out more. So she filed a Freedom of Information Act (FOIA) request to obtain the reports and received a number of ADEs for Nexgard and Bravecto. Dr Carney has generously made these reports available on her website.
ADE Reports For Nexgard And Bravecto
There are specific ADE reports for Nexgard and Bravecto for the period January through March 2016. The results are very similar for both drugs. Dr Carney didn’t get any reports for Simparica as it was too new at the time.
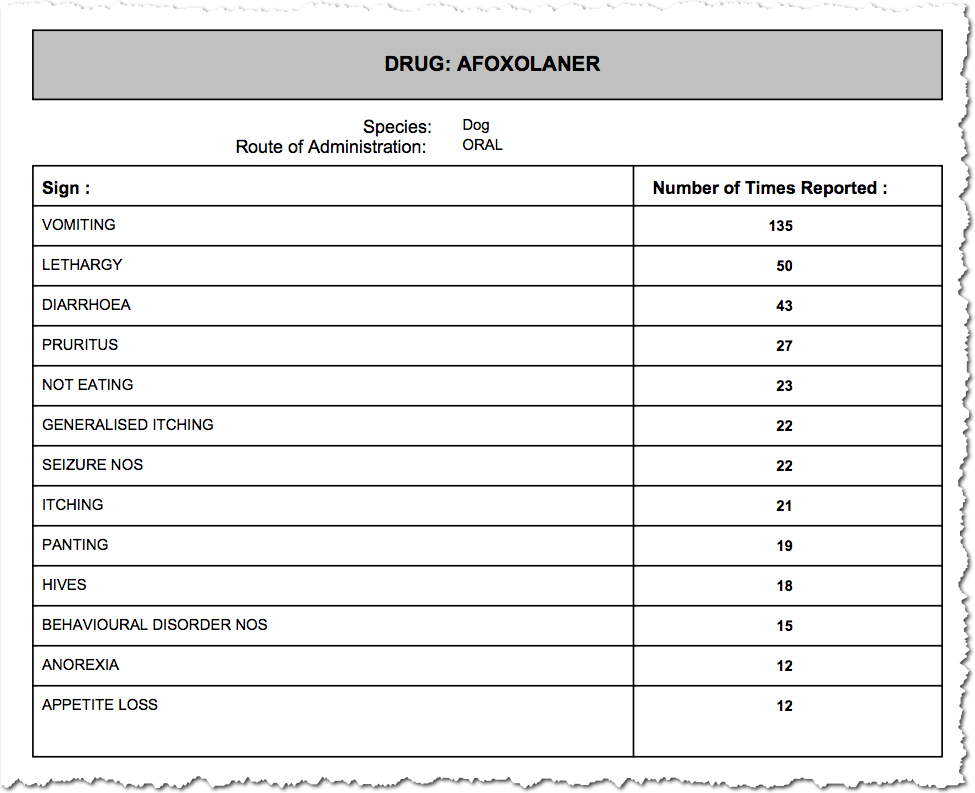
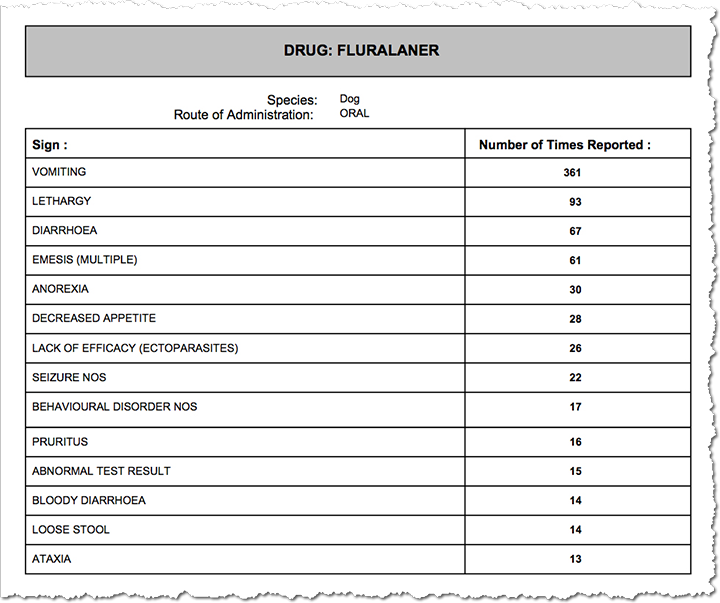
Vomiting, lethargy and diarrhea are the most common side effects reported. Seizures are quite high on both lists, with 22 each for both Nexgard and Bravecto for the first quarter of 2016. Nine deaths are reported for each drug for the same period. For Nexgard, five of the nine deaths were by euthanasia.
Is Nexgard Safe For Dogs?
Is Bravecto Safe For Dogs?
What Dog Owners Say About Chewable Flea and Tick Preventives
Facebook is another source of information about the side effects of these chewable drugs. These group pages contain many tragic stories from dog owners who believe their dogs have been harmed by these pest preventives.
Some of these groups have tens of thousands of members … so the risks are real!
- Does Nexgard Kill Dogs?
- Does Bravecto Kill Dogs?
- Bravecto Nexgard Comfortis Simparica Trifexis & others Do They Kill Dogs?
- Simparica Flea And Tick Medication. Dangerous Drug Causing Seizures (private group)
Some of the posts are fascinating as well as alarming. One poster reports finding dead ticks on her dog as long as 12 months after his last dose of Bravecto … suggesting that’s how long it stays in your dog’s bloodstream!
So again, a reminder that if your dog experiences an adverse effect … you can’t just stop using it and expect your dog to return to normal!
Is Simparica Safe For Dogs?
Simparica hasn’t been out as long … so there’s less information. But the drug works just like Bravecto and Nexgard … it destroys flea and tick nervous systems. So it’s very likely that the adverse effects of this chewable drug are similar to the other two drugs.
The manufacturer’s website provides this warning …
Simparica may cause neurologic signs such as tremors, unsteadiness and/or seizures in dogs with or without a history of neurologic disorders.
If you want to see the kind of effect Simparica can have on your dog, click on this video link. Be warned, though … the video is horrifying to watch. It shows a dog having a really long seizure after taking Simparica. This poor dog died a few months later because of his seizures.
DNM RECOMMENDS: Kin+Kind offers 100% Organic Natural Flea & Tick Spray to protect your dog from pests and soothe itching from bites via topical treatment. Buy Kin+Kind Flea + Tick Prevent! now …
Vets Agree These Drugs Aren’t Safe
What does the veterinary medicine community have to say?
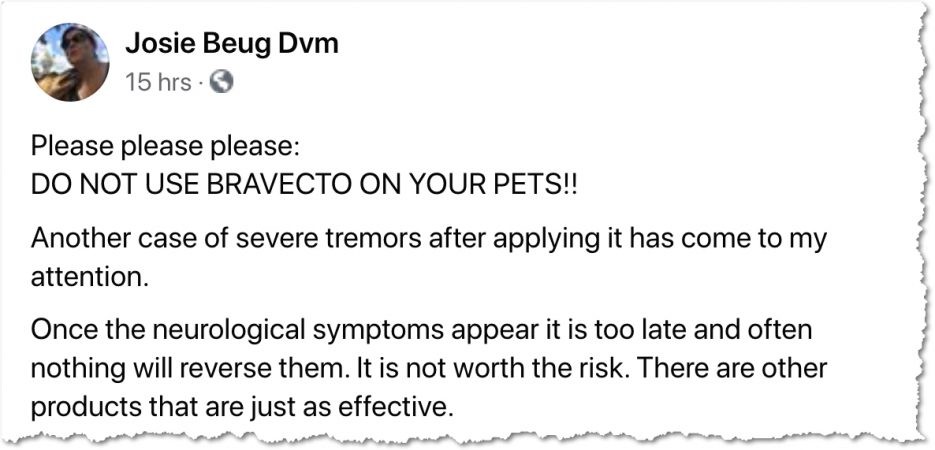
Dr Carney, who obtained these reports from the FDA, told us that she has personally decided not to use these chewable drugs for her own dog, nor will she prescribe them to her clients. Many other holistic vets are speaking out about the dangers of these long-lasting preventive products too. Check out this Facebook post by Dr Josie Beug:
The FDA has also issued Face Sheet about “potential adverse events associatd with Isoxaline Flea and Tick Product.” The FDA specifically warns that “Isoxazoline products have been associated with neurologic adverse reactions, including muscle tremors, ataxia, and seizures in some dogs and cats.“
Please don’t sacrifice your dog’s health for these “convenient” chewable oral preventatives. It’s a really dangerous trade-off, and there are better ways to get additional protection.
The risk of lyme disease and other illnesses makes tick prevention important. But it’s also important to avoid harmful pest preventives for your dog. There are lots of natural, safe options and topical treatments for year round prevention of ticks and adult fleas. You might have to work a little harder … but your dog is worth the effort!
FAQ
What is the best flea and tick preventative?
The best flea and tick preventative avoids harmful chemicals; opt for natural alternatives like Kin+Kind’s 100% Organic Flea & Tick Spray to keep your dog safe from pests without the risk of severe side effects.
What is the best way to prevent fleas and ticks?
The best way to prevent fleas and ticks is by using natural, safe options such as organic flea and tick sprays, maintaining a clean environment, and regularly checking your dog for pests.
What do vets recommend for flea and tick control?
Many holistic vets recommend avoiding chewable flea and tick preventatives due to their potential side effects and suggest natural alternatives like organic sprays and regular grooming for effective pest control.
Is flea and tick prevention worth it?
Yes, flea and tick prevention is worth it to protect your dog from tick-borne diseases, but it’s crucial to choose safe, natural methods rather than potentially harmful chewable medications.












